Biological filtration is the process by which nitrifying bacteria colonized on submerged media surfaces convert ammonia excreted by koi into nitrite, and then nitrite into the far less toxic compound nitrate. It is one of three filtration stages a koi pond system runs simultaneously. Mechanical filtration removes suspended solids before they reach the biological stage. UV clarification targets single-celled algae and certain pathogens. This page covers the biological stage only.
The mechanism driving all of it is the nitrogen cycle. Koi excrete ammonia continuously through their gills and waste. Without a functioning bacterial colony to process that ammonia, it accumulates to lethal concentrations within days. What separates a bio filter that holds a pond in balance from one that fails comes down to three inputs operating at the same time: oxygenated water flow across colonized media, stable water temperature, and a fish load matched to available media surface area. When any one of those inputs breaks down, the chemistry shifts in measurable ways. Every failure mode produces a distinct water test pattern, and knowing which pattern maps to which cause is what turns a test result into a corrective action.
What Biological Filtration Does in a Koi Pond
Biological filtration converts ammonia and nitrite, the toxic byproducts of fish waste, into nitrate using nitrifying bacteria colonized on submerged media surfaces inside the filter. Without an established bacterial colony completing that conversion, ammonia accumulates to lethal concentrations. Koi begin showing measurable physiological stress at 0.5 ppm. At sustained levels above 2 ppm, mortality follows within days (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018).
Mechanical filtration removes suspended solids before they reach the biological stage. UV clarification handles algae and waterborne pathogens through light exposure, not chemistry. Biological filtration does neither. Its job is chemistry conversion performed by living organisms, not by a mechanical or physical process, which means it has no moving parts to inspect and no visible output to confirm it is working. A pump either runs or it does not. A bacterial colony fails silently, and the first sign is usually an ammonia or nitrite reading that should not be there.
The bacteria responsible for nitrification colonize biofilm inside the bio chamber. Biofilm is the thin living layer of bacteria and extracellular material that adheres to submerged media surfaces and provides the colonization substrate the bacterial colony depends on. Nitrosomonas bacteria handle the first conversion step, oxidizing ammonia into nitrite. From there, Nitrobacter and Nitrospira convert nitrite into nitrate, a compound significantly less toxic to koi than either ammonia or nitrite. Nitrate exits the system through partial water changes, plant uptake, or anaerobic denitrification, the breakdown of nitrate into nitrogen gas by bacteria operating in oxygen-free zones. Every step in that sequence depends on the bacterial colony staying alive and adequately supplied with oxygen.
The Nitrogen Cycle in a Koi Pond, Step by Step
Koi excrete ammonia continuously through their gills and waste. Nitrosomonas bacteria convert it to nitrite, Nitrobacter and Nitrospira convert nitrite to nitrate, and nitrate exits through water changes, plant uptake, or denitrification. Understanding what each step consumes is what makes a water test result diagnostic rather than just a number.
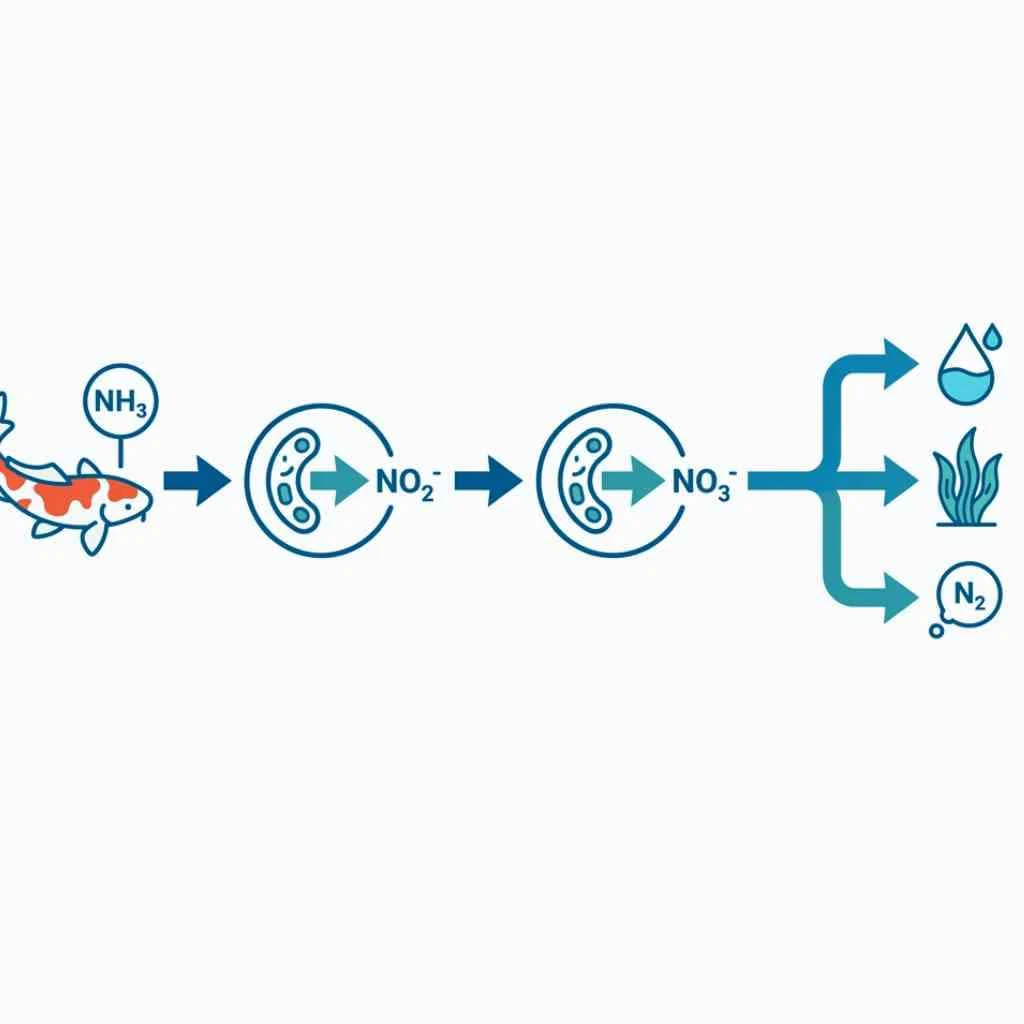
- Ammonia production. Koi excrete ammonia (NH3) continuously through their gills as a byproduct of protein metabolism. Additional ammonia enters the water through uneaten food and decomposing waste. Total ammonia nitrogen (TAN) is the sum of free ammonia (NH3) and ionized ammonium (NH4+). The ratio between the two shifts with pH and temperature, and free NH3 is significantly more toxic to koi than NH4+.
- First-stage nitrification: ammonia to nitrite. Nitrosomonas bacteria oxidize NH3 into nitrite (NO2-). This is the first bacterial conversion step. Nitrosomonas are autotrophic, meaning they derive energy from oxidizing ammonia rather than from organic carbon. They are slow to colonize new media and sensitive to oxygen depletion, pH swings, and chemical disruption.
- Second-stage nitrification: nitrite to nitrate. Nitrobacter and Nitrospira bacteria oxidize NO2- into nitrate (NO3-). Nitrospira is now understood to be the dominant second-stage nitrifier in most mature biofilters, with Nitrobacter playing a secondary role. Nitrate is significantly less toxic to koi than either ammonia or nitrite and does not cause acute mortality at the concentrations typically reached in a managed pond.
- Nitrate removal. Nitrate exits the system through three pathways: partial water changes (the primary method in residential koi ponds), uptake by aquatic plants, and anaerobic denitrification in oxygen-free zones. Most residential koi ponds rely on scheduled water changes as the dominant removal method. Target nitrate concentration for koi is below 40 ppm, with below 20 ppm preferred for heavily stocked systems.
Two consumable inputs run down at every step in this sequence and are rarely tested until a problem appears.
Oxygen demand. Nitrification is an aerobic process. Nitrosomonas consumes 3.16 mg of dissolved oxygen per mg of NH3-N oxidized in the first stage, and Nitrobacter and Nitrospira consume an additional 1.11 mg O2 per mg NO2-N in the second, for a combined demand of 4.57 mg O2 per mg of ammonia-nitrogen fully nitrified (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). When dissolved oxygen at the bio chamber drops below 6 mg/L, nitrification slows. Below 2 mg/L, it stops.
Alkalinity consumption. Nitrification also consumes alkalinity (KH), the carbonate buffer that neutralizes the acids nitrification produces. For every 1 mg of NH3-N oxidized, 7.14 mg of calcium carbonate equivalents (CaCO3) are consumed (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). When KH runs low, those acids accumulate unchecked and pH drops, slowing bacterial activity and shifting the NH3/NH4+ equilibrium toward the more toxic free ammonia form. Correcting a pH crash too quickly after alkalinity exhaustion carries its own ammonia toxicity risk. Raising pH rapidly converts accumulated NH4+ back to toxic free NH3 faster than fish can process the shift.
Where Nitrate Goes: Water Changes, Plants, and Denitrification
Nitrate is removed from a koi pond through three pathways: scheduled partial water changes, uptake by aquatic plants, and anaerobic denitrification in oxygen-free zones. Most residential koi ponds rely on water changes as the dominant removal method. The other two pathways carry significant limitations in a typical koi system.
Water changes are the only removal method a pond owner can control directly and scale to match bioload. A 10–20% weekly water change is the standard management target for residential koi ponds. The volume exchanged matters less than consistency. A pond that receives a 10% change every week maintains more stable nitrate levels than one that receives a 30% change once a month, because nitrate accumulates continuously and irregular large changes create the chemistry swings that stress fish.
Aquatic plants absorb nitrate through their root systems as a nitrogen source for growth. Water lilies, water hyacinth, and watercress are the most commonly cited species for nitrate uptake in koi pond contexts. Capacity is the limiting factor. In a stocked residential pond, plant uptake accounts for a small fraction of total nitrate production relative to what scheduled water changes remove. Plants contribute to nitrate management but cannot replace water changes in any stocked residential pond.
Anaerobic denitrification converts nitrate into nitrogen gas through bacterial activity in oxygen-free zones, and it is the only pathway that permanently eliminates nitrogen from the system. The same oxygen-free conditions that support denitrifying bacteria, primarily Pseudomonas and Paracoccus species, also support anaerobic bacteria that produce hydrogen sulfide, which becomes acutely toxic to koi at concentrations above 0.002 mg/L, a threshold easily reached in an enclosed anaerobic zone (Boyd, Water Quality in Ponds for Aquaculture, Auburn University, 1990). Intentionally engineering anaerobic denitrification into a residential koi pond system introduces a management risk that most residential koi pond systems are not designed to manage.
The management target for dissolved nitrate in a koi pond is below 40 ppm, with below 20 ppm preferred for heavily stocked systems. Research on Cyprinus carpio documents measurable cortisol stress responses at nitrate concentrations well below acute toxicity thresholds, indicating physiological stress that may not present as visible behavioral symptoms (Lupica et al., Aquaculture Research, 2010). Keeping nitrate below the management target reduces that chronic stress load.
How Long a New Bio Filter Takes to Cycle
A new bio filter cycles in 4–6 weeks at water temperatures between 70–75°F. Below 60°F, cycling slows to 8–12 weeks or stalls entirely. Both ammonia and nitrite must read zero ppm. Nitrate detectable in the water confirms the cycle is complete.
Nitrifying bacteria are among the slowest-reproducing organisms used in any biological treatment system. They reproduce by binary fission but do it far more slowly than heterotrophic bacteria because nitrification yields very little energy per reaction. Under optimal conditions at 77–86°F, Nitrosomonas can double every 7–10 hours and Nitrobacter every 13 hours. In a real pond environment, realistic doubling time for the nitrifying colony runs 15–24 hours (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). That doubling time sets the biological rate limit on colonization, which no management intervention fully overrides.
The ammonia spike appears first. As the colony begins to establish, fish waste loads the water with ammonia faster than the small initial Nitrosomonas population can process it. Ammonia climbs, peaks, then drops as Nitrosomonas multiplies to a population capable of keeping pace with the bioload. Nitrite is next. It accumulates as Nitrosomonas output outruns the still-developing Nitrobacter and Nitrospira colony, and it is more acutely lethal to koi than ammonia at equivalent concentrations because it enters the bloodstream and blocks oxygen transport. The cycle is not complete until the nitrite spike also resolves to zero.
Cold water extends every part of this timeline. Below 60°F, bacterial reproduction rate drops by more than half relative to the 77°F baseline, and the entire colonization sequence stretches proportionally. Below 50°F, nitrification effectively stalls. A pond started in early spring in a cool climate may not complete its initial cycle until water temperatures stabilize above 60°F consistently, which means adding koi in early spring to an uncycled system consistently produces ammonia accumulation before the colony can keep pace.
Bacterial seed products are bottled or dry cultures of nitrifying bacteria sold for pond and aquarium use. Dosed correctly by pond gallons rather than aquarium gallons, they start the process with a larger initial colony and can reduce cycling time by 30–50%. The wait is shorter, not eliminated. A seeded pond at 75°F still needs a minimum of 2–3 weeks before the colony is large enough to process a full fish load without ammonia or nitrite accumulating between tests.
When the pond has no fish yet, fishless cycling avoids exposing koi to the ammonia and nitrite peaks that occur while the colony establishes. A chemical ammonia source doses the filter and drives colonization on a controlled schedule. Fish-in cycling applies when koi are present in a system that has not yet completed its initial cycle. It requires daily water testing and partial water changes to keep ammonia and nitrite below acutely toxic thresholds throughout the process.
Cycling Timeline by Water Temperature
Cycle duration ranges from 3–5 weeks at 75–85°F to fully stalled below 55°F. Bacterial activity scales directly with water temperature across the entire range.
Four temperature bands cover the range a koi pond encounters through a typical seasonal cycle. The Q10 temperature coefficient for nitrifying bacteria governs the rate reduction at each band (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018; Sallenave, NMSU Extension, Publication W104).
| Water Temperature | Cycle Duration | Bacterial Activity Level |
|---|---|---|
| 75–85°F | 3–5 weeks | Peak. Optimal colonization rate. |
| 65–75°F | 5–8 weeks | Active. Rate reduced approximately 30–40% from peak. |
| 55–65°F | 8–12 weeks | Slow. Rate reduced approximately 50–75% from peak. |
| Below 55°F | Stalled | Dormant. Nitrification effectively ceases. |
These durations assume a stable fish load throughout the cycle. A pond stocked incrementally as the cycle progresses will cycle faster because ammonia input matches the developing colony size more closely than a pond loaded at full capacity from day one.

Temperature during cycling is rarely stable. A pond that starts at 75°F in late summer and drops to 60°F by week three will not complete its cycle in 3–5 weeks. Bacteria respond to current temperature. The table rows are bands, not guarantees, and a pond crossing from one band into a cooler one mid-cycle resets its effective timeline.
Signs the Cycle Is Complete
The cycle is complete when ammonia reads 0 ppm and nitrite reads 0 ppm for seven consecutive days, nitrate is detectable and rising, and biofilm is visible on media surfaces. Hitting zero once is not confirmation. Seven consecutive days of zero ammonia and zero nitrite means the colony is large enough to process the pond’s current bioload continuously, not just on a good day.
Four indicators confirm cycle completion:
- Ammonia at 0 ppm for seven consecutive days. A single zero reading means little. The bacterial colony must sustain zero ammonia for a full week under normal feeding conditions before the pond is ready for additional fish or increased feeding. Test at the same time each day. Morning tests before feeding give the most conservative reading.
- Nitrite at 0 ppm for seven consecutive days. Nitrite resolves after ammonia. The second-stage colony establishes later and more slowly than Nitrosomonas, and a pond can show zero ammonia while nitrite is still present. Both must hold at zero simultaneously for seven days before the cycle is confirmed complete.
- Nitrate detectable and rising. Nitrate accumulation confirms the full two-stage conversion is running. A pond with zero ammonia and zero nitrite but no detectable nitrate has not completed the cycle. It has either stalled or the ammonia source is too low to drive colonization. Nitrate should be rising between weekly tests, not holding flat.
- Biofilm visible on media surfaces. Established media carries a visible brown or tan coating. That coating is the biofilm layer housing the bacterial colony. Clean-looking media after six or more weeks of operation indicates the colony has not established, the media surface is incompatible, or flow through the bio chamber is bypassing the media.
Aquarium-grade starter products contain the correct bacterial species for koi ponds but are labeled for systems under 150 gallons at low bioload. The dosing gap matters. A koi pond running 500 gallons or more at moderate stocking requires a dose far above the aquarium label recommendation to deliver a meaningful initial colony. Pond-specific products are formulated and labeled for higher-volume, higher-bioload systems.
Use aquarium-grade products only if the pond-specific formulation is unavailable, and multiply the labeled dose by the ratio of pond gallons to the product’s rated tank capacity. Live bacterial cultures lose viability over time, so confirm the product has not exceeded its shelf life before dosing.
Biological Media Types and Surface Area Requirements
Bacterial colony size is limited by media surface area. Static media like lava rock provides an estimated 50–200 ft²/ft³ of total surface area under field conditions, while moving bed media such as K1 and K3 provides 290–415 ft²/ft³ of protected surface area. Sizing depends on fish weight and feeding rate, not pond volume alone.
The distinction between total surface area and protected surface area determines actual bacterial capacity. Total surface area is the manufacturer’s rated figure covering every internal pore and external face of the media. Protected surface area is the portion that water actually reaches under flow conditions, where bacteria can colonize and remain attached without being scoured off. That gap is large on static media.
Ceramic rings, bio balls, matting, and sintered glass all carry manufacturer claims far above their actual performance. Independent testing consistently places the effective surface area of ceramic rings and sintered glass at 15–40 ft²/ft³ because most of the claimed internal pore structure does not receive adequate water flow (Aquarium Science, filter media testing database). Lava rock and matting perform in the same range. The pores and fibers are real but water moves around the media, not through it at the depth the nameplate rating assumes.
Static media loses available surface area through two additional mechanisms over time. Compaction reduces the space between pieces and channels water through the path of least resistance rather than across the full colonized surface. Both are operating failures, not material failures. Organic sludge accumulation from inadequately prefiltered water coats the media and physically blocks ammonia diffusion to the bacterial biofilm beneath.
Moving bed media operates on a different principle. K1 and K3 pieces float and tumble continuously in an aerated water column. Abrasion between pieces strips the outer dead zone of the biofilm and exposes fresh active surface continuously. This is why moving bed media delivers more active bacterial surface per cubic foot than static media at equivalent volume. K1 Micro carries a verified protected surface area of 289.56 ft²/ft³ and K+ carries 313 ft²/ft³ protected with a total of 412 ft²/ft³ (Evolution Aqua product specifications). At manufacturer-rated loads, 50 liters of K1 handles up to 250 grams of food per day and K3 handles up to 225 grams per day (Evolution Aqua, product specification).
The practical sizing rule for moving bed media is 1 lb of fish per gallon of media volume. This applies at moderate summer feeding rates of 1–2% of body weight per day. Heavily stocked ponds or ponds fed at higher rates require proportionally more media. A second bio chamber is the correct response to outgrown capacity, not operating the existing volume beyond its rated load.
Specific Surface Area Compared by Media Type
Moving bed media delivers the highest protected surface area per cubic foot because continuous abrasion exposes fresh colonization sites and overcomes the mass transfer limitation that caps mature biofilms on static media.
| Media Type | Total Surface Area (ft²/ft³) | Protected Surface Area (ft²/ft³) | Self-Cleaning | Best Use |
|---|---|---|---|---|
| Lava rock | ~50–200 | Low. Effective area limited by water flow around, not through, the rock | No | Low-cost static bed where volume is not constrained |
| Plastic bio balls | ~100–200 | Low. Outer surface only; inner structure receives minimal flow | No | Trickle/shower filters where surface exposure to air compensates |
| Ceramic rings / sintered glass | ~15–40 effective despite high nameplate claims | Low. Internal pore structure does not receive adequate flow under field conditions | No | Small-volume filters where space limits media volume |
| Matting | ~50–150 | Moderate when clean; drops significantly as organic load accumulates | No | Mechanical pre-filtration stage rather than primary bio media |
| K1 / K3 moving bed | 290–415 total | 289–313 verified protected (Evolution Aqua product specifications) | Yes | Primary bio chamber in koi pond systems at any scale |
The mechanism behind the protected surface area gap on static media is biofilm mass transfer limitation. As biofilm accumulates on static media surfaces, the outer heterotrophic layer thickens and blocks oxygen and ammonia diffusion to the active nitrifying bacteria deeper in the biofilm. The diffusion coefficient of oxygen inside a mature biofilm runs at 40–80% of its value in free water (Gujer and Boller, Water Science and Technology, 1986).
That means the bacteria furthest from the surface operate in an oxygen-depleted environment regardless of dissolved oxygen levels in the water column, a finding confirmed across biofilm reactor literature including Timmons, Guerdat, and Vinci (Recirculating Aquaculture, 4th ed., 2018). The nameplate surface area remains unchanged. The active bacterial capacity does not.
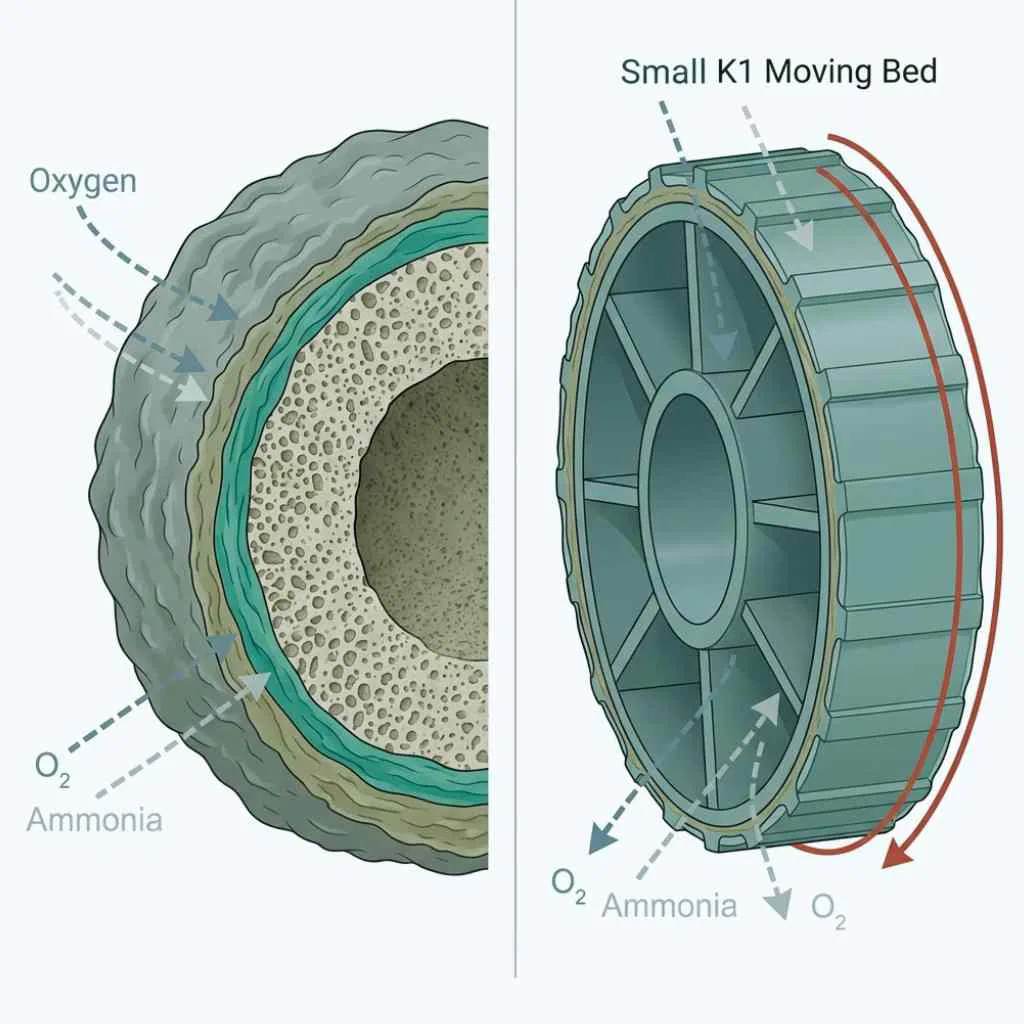
Moving bed abrasion resolves this directly. The tumbling action strips the outer dead layer of the biofilm continuously, keeping the active biofilm thin enough for oxygen and ammonia to diffuse through to the nitrifying bacteria.
How to Calculate Media Volume Needed for Your Bioload
Calculate media volume in five steps: estimate total fish weight, determine peak summer feeding rate, apply the media’s protected surface area rating against the 100 ft² per lb of fish standard, add a 25% safety margin, and compare the required volume to your current media volume.
- Estimate total fish weight in pounds. Weigh each fish if possible. For koi, length is a reliable proxy: 12 inches weighs approximately 0.5 lbs, 18 inches approximately 1.5–2 lbs, and 24 inches approximately 4–6 lbs depending on body depth. Add individual weights for the total. A filter sized to current fish weight will be undersized within 12 months as koi continue growing, so estimate at the weight they will reach within the next 12 months, not current weight.
- Determine peak summer daily feeding rate. Use 1.5% of total body weight per day as the baseline calculation figure for a moderately fed pond. That is the standard. A pond carrying 20 lbs of koi at 1.5% receives 0.3 lbs (136 grams) of food per day at peak summer feeding.
- Apply the 100 ft² per lb of fish standard to your media’s protected surface area. The established sizing standard is 100 ft² of effective surface area per pound of fish (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). A 20 lb fish load requires 2,000 ft². K1 Micro provides 289.56 ft²/ft³ of protected surface area. Divide 2,000 by 289.56: 6.9 cubic feet required. Multiply by 7.48 to convert: approximately 51.6 gallons of media volume.
- Add a 25% summer safety margin. Peak summer conditions stack maximum feeding rates, elevated water temperature, and reduced oxygen solubility at the same time. Multiply the Step 3 volume by 1.25. For the example above, 51.6 gallons becomes 64.5 gallons required.
- Compare required volume to current media volume. Measure the current media volume in your bio chamber. If current volume falls below the Step 4 figure, the system is operating below its surface area requirement at peak summer load. Operating existing media volume above its rated capacity does not increase bacterial surface area and does not resolve the deficit. The correction is upgrading media type or adding a second bio chamber.
Why Oxygen Is the Hidden Bottleneck in Bio Filtration
Nitrifying bacteria consume 4.57 mg of dissolved oxygen for every 1 mg of ammonia they oxidize (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). When dissolved oxygen at the bio chamber drops below 6 mg/L, nitrification slows measurably. Below 2 mg/L, it stops entirely. Most bio filters that fail despite adequate media volume are failing because of inadequate oxygen delivery, not inadequate surface area.
Oxygen solubility in freshwater decreases as water temperature rises. At 60°F, freshwater at saturation holds approximately 10.0 mg/L of dissolved oxygen. At 80°F, that figure drops to approximately 8.1 mg/L, a reduction of roughly 19% (Benson and Krause solubility values, USGS Water Quality Technical Memorandum 2011.03). A pond running well in spring can cross below the nitrification threshold at the bio chamber by midsummer with no change in media, pump flow, or fish load. In ponds deeper than 3 feet, summer temperature stratification can form a warm low-oxygen surface layer that the bio chamber pump draws from, reducing delivered oxygen below the saturation value the surface reading suggests.
Bacteria and fish draw from the same oxygen pool. Fish extract oxygen across their gills in proportion to metabolic rate. Nitrifying bacteria extract it from the water flowing across the biofilm surface. Both demands peak simultaneously in summer. Organic matter decomposition adds a third oxygen draw through biological oxygen demand (BOD), the load created by heterotrophic bacteria breaking down uneaten food and waste in the same water column. The competition is not visible until a water test shows ammonia that should not be there given the installed media volume.
Trickle filters and shower filters outperform submerged bio chambers per cubic foot of media in warm weather. The mechanism is oxygen delivery: the water is exposed to air during nitrification rather than depending on dissolved oxygen carried in from the rest of the system. A submerged bio chamber is limited by the dissolved oxygen concentration of the water entering it. A trickle tower re-oxygenates continuously as water passes through the media.
Supplemental aeration at the bio chamber is the highest-return fix for a stalled filter where media volume is already adequate. A diffuser stone placed at the chamber inlet raises dissolved oxygen before the water reaches the bacterial colony. This pattern appears consistently in summer diagnosis: adequate media, adequate flow, ammonia present, and dissolved oxygen at the bio chamber running below 5 mg/L. When oxygen limitation is the cause, adding aeration typically reduces ammonia within days without any change to media or flow.
Two symptoms indicate oxygen-limited nitrification before water testing confirms it. Ammonia present despite media volume that tested adequate at a cooler water temperature is the primary indicator. Fish gasping at the surface in early morning, before photosynthesis has added oxygen to the water column, means the oxygen deficit has moved beyond the bio chamber and is affecting the whole pond.
How Water Temperature Controls Filter Performance
Nitrifying bacteria activity halves with every 18°F (10°C) drop below 77°F. Below 50°F, nitrification stops. Spring is the highest-risk window because koi resume feeding before the bacterial colony reactivates.
The optimal nitrification range is 77–86°F, where bacterial reproduction and ammonia oxidation run at peak rate. Activity declines in proportion to temperature drop below that range, governed by the Q10 temperature coefficient, the biological rate constant describing how reaction rates change with a 10°C shift in temperature (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). Below 50°F, activity effectively ceases. Koi feeding rate follows a graduated scale tied directly to water temperature: minimal acceptance below 50°F, once every two to three days at 50–55°F, once daily at 55–65°F, and full feeding above 65°F as metabolism fully supports digestion.
Koi metabolism and feeding behavior restart at 50–55°F, driven by water temperature alone. The bacterial colony does not restart on the same schedule. At 50°F, koi will accept food if offered. Nitrosomonas and Nitrospira are near-dormant at that same temperature. The mismatch between feeding resumption and bacterial reactivation creates a 2–4 week spring window where ammonia accumulates faster than the filter can process it.
Feeding management reduces the spring ammonia risk window. At water temperatures below 55°F, reduce feeding to once every two to three days using a wheat germ-based or low-protein cold-water formula. Protein-heavy food produces significantly more ammonia per gram fed than cold-water formulas. Koi cannot fully digest protein at low temperatures, and the undigested portion passes as waste that loads the water column directly. A pond where standard feeding resumes at 55°F will typically see ammonia climb within three to five days, not because the filter has failed, but because the bacterial load capacity has not yet matched the feeding rate.
The bacterial colony does not restart uniformly. Nitrosomonas reactivates faster than Nitrobacter and Nitrospira because ammonia accumulates first and drives first-stage colonization before nitrite builds. A spring nitrite spike follows: first-stage nitrification resumes before second-stage, nitrite accumulates briefly, then resolves as water warms and the second-stage colony catches up. That sequence typically completes within one to two weeks of the ammonia spike resolving.
The Six Most Common Bio Filter Failure Modes
Bio filter failure shows up in water tests before fish show symptoms. Each test pattern points to a specific cause: oxygen limitation, second-stage colony lag, prefilter neglect, alkalinity loss, chemical kill, or hydraulic dead zones. Identifying which failure mode is present requires a water test. In field diagnosis, visual inspection of the fish or the filter alone frequently misidentifies the cause because most failure modes produce identical behavioral symptoms in early stages.
Koi showing reduced appetite, clamped fins, or surface respiration could be responding to elevated ammonia, elevated nitrite, low pH from alkalinity crash, or dissolved oxygen depletion. All four conditions look the same on the fish. They do not look the same on a water test. Ammonia present with nitrite zero points to a different failure than ammonia and nitrite both present. pH below 6.5 with rising ammonia points to a different failure than normal pH with rising ammonia. The test result is the diagnosis.
Water testing must cover four parameters to diagnose bio filter performance accurately: ammonia, nitrite, pH, and KH (carbonate hardness). Nitrate confirms the full two-stage conversion is running but does not change the immediate diagnostic picture. Dissolved oxygen at the bio chamber is the diagnostic reading, not the surface. A pond owner testing at the surface and not at the bio chamber inlet can miss the oxygen limitation failure mode entirely while the filter runs at suppressed capacity.
Corrective action depends on correctly identifying which failure mode is present first. Misidentifying it compounds the problem. Treating an oxygen limitation failure as a media volume problem leaves the oxygen cause unresolved while adding cost and delay.
Ammonia Present but Nitrite Zero
Ammonia present with nitrite zero means Nitrosomonas colonization is failing or absent. The most common cause is oxygen limitation at the bio chamber. The second most common is a filter that has not yet completed colonization following a new build, recent cleaning, or chemical treatment.
Symptom. The pattern is ammonia above 0.25 ppm with nitrite at zero ppm. Nitrite zero with rising ammonia confirms the first nitrification stage is not converting. If nitrite were also elevated, that would indicate a different failure mode with both stages under stress.
Diagnostic test. Measure dissolved oxygen at the bio chamber outlet, not at the water surface. A surface reading above 6 mg/L does not confirm adequate oxygen at the bacterial colony. Check whether the filter was cleaned, restarted, or chemically treated within the last 30 days. A prior event within that window is the most likely explanation if the pond was previously stable.
Root cause. Oxygen below 6 mg/L at the bio chamber suppresses Nitrosomonas activity before any other visible symptom appears. Insufficient oxygen is the primary diagnosis when no recent cleaning or chemical event occurred. If a cleaning or chemical event happened within 30 days, the colony is in the pre-colonization or recovery stage.
Corrective action. Add aeration directly to the bio chamber inlet or base. Confirm dissolved oxygen at the chamber outlet reaches 6 mg/L or above. If the cause is a recent cleaning event rather than oxygen limitation, allow 4–6 weeks for re-colonization at current water temperature, adjusting upward if water is below 65°F. Perform a 20% water change to reduce the ammonia load while the colony recovers, and reduce feeding to once daily until ammonia holds at zero for three consecutive days.
Both Ammonia and Nitrite Present
Ammonia and nitrite both present simultaneously indicates the bio filter is overloaded and bacterial capacity is insufficient to process the current bioload. The cause is overstocking, overfeeding, a sudden bioload increase from new fish additions, or partial colony die-off from a recent stressor.
Symptom. The pattern is ammonia above 0.25 ppm and nitrite above 0.25 ppm at the same time. Both nitrification stages are falling behind simultaneously. This pattern is more acutely dangerous than ammonia-only because nitrite converts hemoglobin to methemoglobin, which cannot carry oxygen.
Diagnostic test. Count fish and estimate total fish load. Check for recent fish additions or feeding rate increases within the last two to four weeks. Calculate daily feeding rate as a percentage of total fish weight and compare against the media volume sizing standard. Measure pH and KH to rule out an alkalinity crash running alongside the capacity overload, since both conditions produce rising ammonia simultaneously.
Root cause. Bioload exceeds the bacterial colony’s current processing capacity. Both stages are operating below the load they are receiving. The first-stage colony cannot convert ammonia to nitrite as fast as it is produced, and the second-stage colony falls behind at the same rate.
Corrective action. Stop feeding for 48–72 hours. Perform a 20–30% water change to dilute both ammonia and nitrite. Add non-iodized salt at 0.1–0.3% to raise chloride levels. Chloride ions compete with nitrite for uptake sites on the gill carrier mechanism, reducing nitrite toxicity while the filter recovers (Boyd, Global Aquaculture Advocate, 2014; Masser et al., SRAC Publication No. 0462, 1992). If the cause is permanent overstocking rather than a transient event, upgrade media volume or add a second bio chamber to match the actual fish load.
Nitrite Spike After Cleaning the Filter
A nitrite spike within 1–2 weeks of cleaning the filter means the bacterial colony was disrupted by the cleaning event. The second-stage colony recovers slower than the first. Nitrobacter and Nitrospira recolonize after Nitrosomonas, producing the nitrite accumulation pattern that follows a cleaning event.
Symptom. The pattern is nitrite rising 3–14 days after a filter cleaning, water change, or media replacement. Ammonia may be low or zero if the first-stage colony recovered quickly. The timing is the diagnostic: nitrite elevation with no other system change in the preceding two weeks points directly to the cleaning event.
Diagnostic test. Confirm whether bio media was rinsed under running tap water rather than pond water. Check whether media was scrubbed aggressively or replaced in full. These actions remove biofilm physically and reduce the colonized surface available to the bacterial colony.
Root cause. The primary mechanism is physical disruption. Running bio media under tap water for 30–60 seconds dislodges the biofilm layer that houses the bacterial colony, regardless of chlorine concentration. Soaking media in chloraminated tap water adds a secondary kill risk, as chloramine at residential concentrations is more stable and more damaging to bacteria than free chlorine under prolonged contact (Cunliffe, Applied and Environmental Microbiology, 1991). Aggressive scrubbing removes biofilm more thoroughly than rinsing.
Corrective action. Rinse bio media in pond water only. Clean prefilter and bio media on separate days. Replace no more than 25% of bio media at one time. Add a bacterial seed product after any cleaning event. If a nitrite spike is already present, reduce feeding to once daily and perform a 20% water change while the second-stage colony recovers.
pH Crash Killed the Colony
A pH crash below 6.0 kills nitrifying bacteria and stalls the entire nitrogen cycle. The crash happens because nitrification consumes alkalinity (KH), the carbonate buffer that neutralizes the acids the bacterial colony produces. When KH falls below 2 dKH, the buffering system becomes insufficient and pH can drop overnight.
Symptom. The pattern is pH below 6.5 with rising ammonia and a KH reading below 2 dKH. Both readings together confirm the failure. pH below 6.5 alone has other causes. The combination of pH below 6.5, KH exhaustion, and rising ammonia points directly to alkalinity-driven colony collapse.
Diagnostic test. Measure pH at sunrise and again at sunset on the same day. Diurnal pH swings greater than 0.5 between the two readings indicate KH exhaustion, because a healthy carbonate buffer holds pH stable across the daily photosynthesis-respiration cycle. Measure KH directly. A reading below 2 dKH confirms the buffer is insufficient.
Root cause. Nitrification consumed alkalinity faster than it was replaced. Every milligram of ammonia-nitrogen oxidized consumes 7.14 mg of calcium carbonate equivalents (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). That depletion removes the buffer. CO2 from biological respiration then accumulates overnight when photosynthesis stops, driving pH further down into the range where bacterial activity ceases.
Corrective action. Raise KH using non-iodized baking soda: 1 tablespoon per 100 gallons raises KH by approximately 1 dKH. Add incrementally over 24–48 hours. Target KH 5–8 dKH.
Parallel risk. Correcting pH after a crash carries its own ammonia toxicity risk. At low pH, total ammonia nitrogen (TAN) exists predominantly as ionized ammonium (NH4+), which is significantly less toxic to koi than free ammonia (NH3). The equilibrium shifts. As pH rises during correction, accumulated TAN converts toward the more toxic NH3 form. Raise pH by no more than 0.2 units per day and test ammonia at each step. If TAN is above 1 ppm before correction begins, perform a 20–30% water change first.
Antibiotic or Medication Wiped the Filter
An ammonia spike following pond medication points to chemical disruption of the bacterial colony. The medications that pose the highest documented risk to an established bio filter are antibiotic bath treatments and methylene blue. The distinction matters. Formalin, malachite green, and copper sulfate at standard therapeutic doses show minimal effect on established filters in controlled studies.
Symptom. The pattern is ammonia rising 3–7 days after pond medication. Nitrite may follow within a week as the damaged first-stage colony output drops. The timing window between medication and the ammonia spike is the primary diagnostic indicator.
Diagnostic test. Confirm what was added to the pond and at what dose. Check whether treatment was administered directly to the main pond or in a separate quarantine tank. Confirm whether activated carbon was added after treatment to remove residual medication. Note the duration of treatment, since repeated or prolonged exposure increases disruption risk beyond a single dose.
Root cause. Antibiotic bath treatments, including erythromycin and other aminoglycosides, reduce biofilter function by 44–100% depending on concentration and colony maturity (Collins et al., 1976; IFAS FA100). Methylene blue at 5 mg/L causes complete cessation of nitrification for up to 16 days (Levine and Meade, 1976; IFAS FA100). Both compounds are documented filter risks. An immature or recently established filter is more vulnerable to any chemical stressor, including formalin and copper, than a mature filter with established biofilm.
Corrective action. Treat fish in a quarantine tank rather than the main pond. Treating in a quarantine tank removes the main pond filter from all chemical exposure. Run activated carbon to remove residual medication, then perform a 30–50% water change. Add a bacterial seed product and reduce feeding to near-zero for two weeks while the colony recovers. Salt at standard pond concentration and praziquantel at label dose do not harm the bio filter and are the preferred treatment options when quarantine is not available.
Channeling Through Static Media
Channeling is when water carves preferential paths through static media, bypassing most of the colonized surface area. The bio filter performs at a fraction of its rated capacity even when media volume is correct. A pond owner who added media to address an ammonia problem and saw no improvement is frequently dealing with channeling rather than insufficient volume.
Symptom. The pattern is ammonia or nitrite present despite adequate media volume and dissolved oxygen levels that test within range. Visible water tracks run across the surface of the media bed rather than distributing evenly. Sludge accumulates in some pockets while other zones look clean, indicating water has been avoiding those zones entirely.
Diagnostic test. Inspect the bio chamber surface while the pump is running. Visible flow concentration confirms channeling. Lift media samples from multiple zones across the chamber. Colonized media carries a brown or tan biofilm coating. Clean-looking media in a system running for months indicates that zone has not been receiving adequate flow.
Root cause. Uneven flow distribution is the most common initiating cause. A single inlet point drives water through the path of least resistance, and media compaction reduces void space between pieces over time. Both narrow the effective flow path. Sludge accumulation from inadequate prefiltration fills remaining void space and directs flow further away from colonized zones, starving that portion of the colony of ammonia and oxygen simultaneously.
Corrective action. Rinse media in pond water to break up compacted channels. Redistribute media evenly across the chamber floor. Confirm the inlet delivers flow across the full media bed width rather than concentrating at one point. Install a flow distribution plate if the chamber geometry concentrates incoming flow. For systems that channel repeatedly despite redistribution, convert from static media to K1 or K3 moving bed, which self-distributes through abrasion.
Mechanical Prefiltration Protects the Bio Filter
Solids reaching the bio chamber smother bacterial colonies, consume the oxygen bacteria need to nitrify, and create channeling through the media. A bio filter without effective mechanical prefiltration progressively loses capacity as organic matter accumulates in the media bed.
Solids that bypass the prefilter decompose inside the bio chamber. Decomposition is aerobic. It competes directly with nitrifying bacteria for the same dissolved oxygen pool, and as decomposition products accumulate, void space reduces, channeling develops, and effective colonized surface area shrinks.
Dissolved organic carbon (DOC), the fraction of organic matter that passes mechanical filtration in solution rather than as particles, feeds heterotrophs directly in the bio chamber and requires foam fractionation or protein skimming to remove.
Heterotrophic bacteria pose the more damaging long-term threat to nitrifier capacity when organic load is high. Heterotrophs have a maximum growth rate approximately five times higher than autotrophic nitrifiers (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018). When organic load is high inside the bio chamber, heterotrophs reproduce rapidly and physically displace nitrifying bacteria from colonized surface area. The nitrifiers are not killed. They are outcompeted for attachment sites.
Sludge in the bio chamber is not a bio filter problem. It is a prefilter problem that has migrated downstream. If the bio chamber has visible sludge, the prefilter is undersized, under-maintained, or both.
Prefilter type determines the maintenance schedule required to keep organic load out of the bio chamber. Brush and matting prefilters require weekly cleaning at normal koi pond bioload. Drum and sieve filters are self-cleaning or require daily attention at high bioload. Settlement chambers and vortex units require solids removal every two to three days. Delayed cleaning allows organic load to pass into the bio chamber and extend the window during which heterotrophs outcompete nitrifiers for surface area.
Turnover Rate and Flow Behavior Through the Bio Chamber
A koi pond bio filter needs full pond turnover every one to two hours. Faster than that reduces bacterial contact time. Slower starves the colony of ammonia and oxygen. Flow distribution across the media surface matters as much as total flow rate.
The one to two hour turnover target applies to the bio filter specifically, not to the pump nameplate rating. Head loss across mechanical filters, UV units, plumbing bends, and elevation changes can cut actual flow well below the pump’s rated output. Measure actual flow through the bio filter by timing pond drawdown at the outlet rather than the pump inlet, since head loss accumulates between the pump and the bio chamber and the outlet reading reflects what the bacteria actually receive (Timmons, Guerdat, and Vinci, Recirculating Aquaculture, 4th ed., 2018).
Contact time is the variable that turnover rate controls. Nitrifying bacteria in a biofilm require a minimum contact period to transfer ammonia across the concentration gradient into the biofilm. At flow rates above approximately three times pond volume per hour, water passes the colonized surface faster than the bacteria can process the load. At the lower bound, flow below one pond volume per hour reduces both ammonia delivery and dissolved oxygen concentration at the biofilm surface, suppressing nitrification through substrate starvation rather than contact time limitation. Conversion efficiency drops progressively as flow rate climbs above two times per hour.
Flow distribution determines whether the full media surface participates in nitrification or only a fraction of it. Uneven distribution creates hydraulic dead zones where bacteria starve of both ammonia and oxygen. This is why two bio chambers with identical media volume can show substantially different nitrification output. The variable is not volume. It is how flow enters and distributes across the media.
Moving bed reactors tolerate variable flow better than static media chambers. Static media chambers operate as plug flow reactors, meaning water moves in a single direction through a fixed bed. When flow is uneven, most of the bed becomes a hydraulic dead zone that receives no substrate. The tumbling action of moving bed media redistributes the colonized surface continuously, eliminating the fixed flow paths that allow dead zones to persist.
A pond can have too much filtration in the hydraulic sense. Flow rates above three times pond volume per hour cut contact time below the bacterial conversion window. Oversized media without matched flow creates the same dead zone problem as undersized flow. The design target is matched sizing across flow rate, media volume, and distribution geometry.
Should the Pond Filter Run 24 Hours a Day
Yes. Run the pond filter continuously. Turning off the pump for more than a few hours starves the bacterial colony of oxygen and ammonia simultaneously. Measurable die-off begins within 6–12 hours at water temperatures above 75°F. Intermittent pump operation produces a filter recovery period that typically exceeds any electricity savings in time, water changes, and fish stress.
Nitrifying bacteria are obligate aerobes. They cannot survive without oxygenated water flow. When the pump stops, water in the bio chamber stagnates and dissolved oxygen drops as the colony consumes what remains. Within hours at warm temperatures, anaerobic conditions at the biofilm surface begin suppressing nitrifying activity. The filter does not pause when the pump stops. Bacterial activity begins declining immediately as dissolved oxygen at the biofilm surface drops.
Restart after a long shutdown does not resume where the filter left off. The disrupted colony must recolonize the media through the same staged sequence as an initial cycle: ammonia spikes as Nitrosomonas re-establishes, then nitrite spikes as Nitrobacter and Nitrospira lag behind. Depending on shutdown duration and water temperature, this mini-cycle takes one to three weeks to resolve. The fish are in the pond throughout.
Two shutdown conditions are legitimate. Short maintenance windows under two hours carry low risk because residual dissolved oxygen sustains the colony during that window. Winter dormancy in regions where the pond surface freezes is valid only when a separate aeration source maintains dissolved oxygen at depth where koi shelter.
For pond owners concerned about electricity costs, the correct response is upgrading to a variable speed or high-efficiency pump rather than cycling the pump on and off. A modern variable speed pump running continuously at optimal flow consumes significantly less electricity than an older fixed-speed pump running the same hours. The pump should not be turned off to save electricity.
When to Repair, Reseed, or Replace a Failing Bio Filter
Reseed when the colony is killed but the system is sized correctly. Repair when one input has failed. Replace when the system can no longer perform within its current design. Each of these decisions requires a specific symptom pattern from a water test, not a visual assessment of the pond or the filter.
Each failure presentation maps to a specific cause and a specific action: Reseed, Repair, or Replace. The distinction between Repair and Replace is not about equipment age. It is whether the failure resolves by correcting one variable or whether the system architecture is the constraint.
| Symptom Pattern | Likely Cause | Action |
|---|---|---|
| Ammonia or nitrite spike within 3–14 days of a medication or cleaning event | Chemical or mechanical colony kill | Reseed. Bacterial starter product plus 30% water change plus reduced feeding for 2 weeks while the colony re-establishes. |
| Persistent ammonia despite media volume that calculates as adequate | Oxygen limitation or channeling | Repair. Add aeration directly to the bio chamber. Redistribute static media and confirm flow distribution across the full media bed. |
| Persistent ammonia with pH below 6.5 and KH below 2 dKH | KH exhaustion and pH crash | Repair. Raise KH to 5–8 dKH using baking soda dosed incrementally. Monitor pH stability over two weeks before increasing feeding. |
| Prefilter overflow or visible sludge in the bio chamber | Undersized or neglected mechanical stage | Repair or Replace prefilter. Upgrade prefilter capacity or cleaning frequency. If the bio colony crashed from organic overload, Reseed after the prefilter issue is resolved. |
| Ammonia present and bioload has increased substantially since the system was designed | Bioload has outgrown filter capacity | Replace media or add a second bio chamber. Size the replacement against the current fish weight and feeding rate at the 100 ft² per lb of fish standard. |
| Multiple simultaneous failures; hydraulics cannot sustain 1x pond turnover per hour; channeling returns after repeated redistribution | System has reached end of functional life | Replace. Full filter system upgrade required. Professional consultation is recommended before specifying replacement components. |
Three conditions consistently indicate that professional pond maintenance is warranted. Multiple simultaneous failure modes where correcting one does not resolve the others. Repeated failure of the same corrective action within weeks of each attempt. A system where pond volume, fish load, or hydraulic geometry has changed substantially since the original filter was specified.
